 
Positions are measured using the meter sticks, then wavelengths are determined from the positions using the graph itself or the equation of the best fit line for that graph.įor atoms that contain only one electron, the theory of atomic structure proposed by Niels Bohr can be used to calculate wavelengths for transitions between particular electronic energy levels of the atom. The calibration graph is therefore an integral part of the spectroscope. Students come up with questions that they would like to answer to connect specific aspects of atomic structure with energetic and. Atomic absorption Spectroscopy, Atomic emission spectroscopy. For example, using the same apparatus and without moving the relative positions of the meter sticks, diffraction grating and lamp, it is possible to view the spectrum of a new element, measure where its spectral lines occur on the meter stick, and then read the graph or use the equation of the line to determine the wavelength to which each of those positions corresponds. Theory examination, the Junior Analyst will become eligible to be declared as Food. Once the best fit straight line has been determined, the equation of this line can then be used to convert positions of other spectral lines to wavelength. position of the spectral line will yield a straight line. In the pre-quantum-mechanical period the general method of working on. Background The Rydberg Formula for the wavelength of the radiation emitted in atomic energy level transitions is 1 Z²R1 - 1) Infinal ninitial where R 1. two states of an atom a line results from the totality of transitions between. Visible light consist of 7 colours Violet. Since this position depends upon the wavelength in a linear way, a graph of wavelength vs. Atomic Spectra And Atomic Structure 12 Pre-Lab Questions Before Beginning This Experiment In The Laboratory, You Should Be Able To Answer The Following Questions. Lab 9 Atomic Line Spectra and PHYS 112 Atomic Structure Name: I. Answer 1: The colour of the visible light beginning with that of highest energy (shortest wavelength) is Violet. Using a light source that contains known wavelengths of light, we can measure exactly where each known wavelength appears along a meter stick. For two questions on the topic of atomic structure which was taught using. As the light emerges after being reflected by the grating, these tiny lines cause the reflected light to interfere with itself in such a way that the different wavelengths of the light to appear in different positions to the left and right of the original direction in which the light was traveling. These results underline the importance of assessment in student uptake of. A diffraction grating is a piece of glass or clear plastic with many very narrow and closely spaced lines on it. If we view the light through a prism or a diffraction grating, however, the individual wavelengths are separated. To the naked eye, the various wavelengths (colors) of light emitted by an element are mixed together and appear as a single color that is a combination of the component colors. To measure these wavelengths in the laboratory, we must first separate them. What is the purpose of this experiment, in the form of a question answer choices. 
Thus, the spectrum of an element can be stated by listing the particular wavelengths of light that its atoms emit. These two relationships combine to give a third: Chemistry questions and answers Rb Atomic Spectra and Atomic Structure Pre-Lab Name Section Numben Dater PRE-LAB QUESTIONS Show all work for full credit. 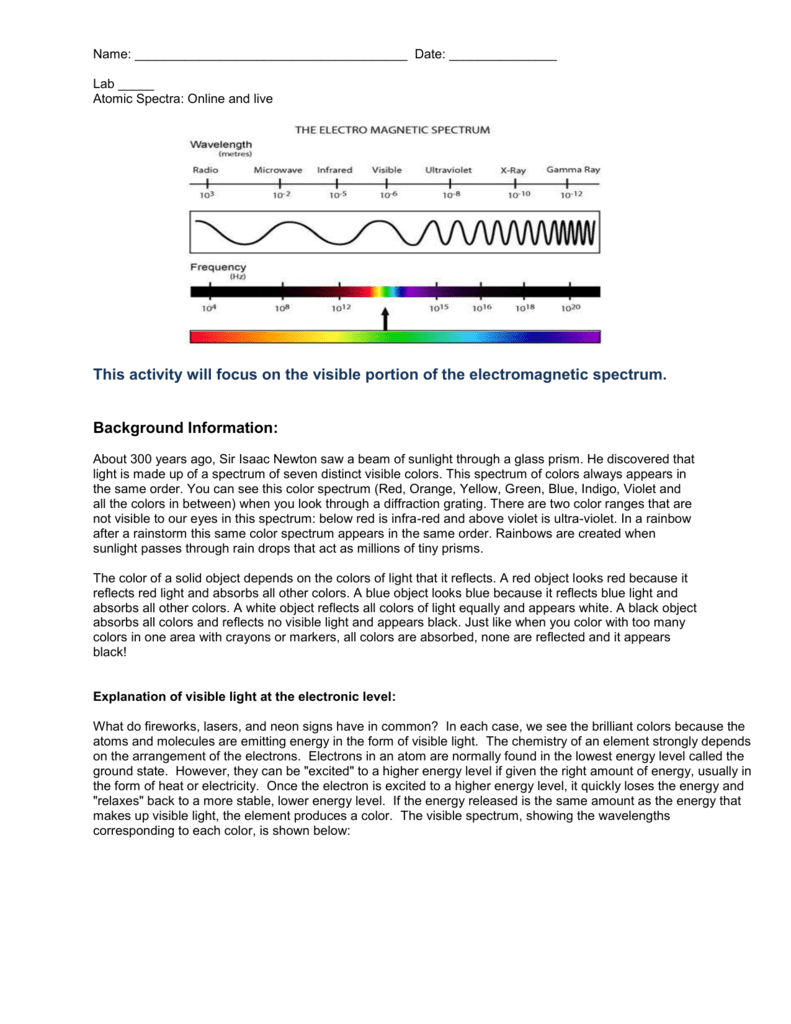
Background The Rydberg Formula for the wavelength of the radiation emitted in atomic energy level transitions is 1 Z²R1 - 1) Infinal ninitial where R 1. Write down the equation of the line (you can get away with using only 4 decimal places) - you will need it for calculations further in the assignments.Js\). Question: Lab 9 Atomic Line Spectra and PHYS 112 Atomic Structure Name: I. 4.Under the next heading ("Graph"), click to make a scatter plot in the 2nd drop-down menu, and click "Show Trend Line" then click refresh. Literature Wavelengths in a Helium Gas Discharge Bulb - A Calibration Curve" in the title box, "Observed Wavelengths (in nm)" in the X-Axis Label box and "Literature Wavelengths (in nm)" in the Y-Axis Label box. Under the data table there is a heading "Graph Settings" with three text boxes. Click the red "x" at the end of the row and delete any rows you don't have an observed wavelength for. Otherwise choose the closest one to your observed value. If your observed wavelength is exactly between two literature wavelengths, you can pick either one. Match up the wavelengths for the lines you saw with their counterpart in the second column and enter your observed wavelengths for helium in the first column of the data table. This is your (Helium) calibration curve: 1.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
